Instead of conducting an experiment this week, we wanted to research more about our industrial process design (lycopene extraction) and expand our PFD from week 2. We also started preparing for our final report on our lycopene extraction simulation.
Lycopene Design PFD
Similar to the PFD in week 2, the equipment we had in the PFD was an extraction tank, storage tank, boiler, and distillation column. However, we needed one more equipment to fit the requirements of the assignment, so we added a tomato washer since it is important to clean bacteria off of them so it doesn’t contaminate anything during the process.
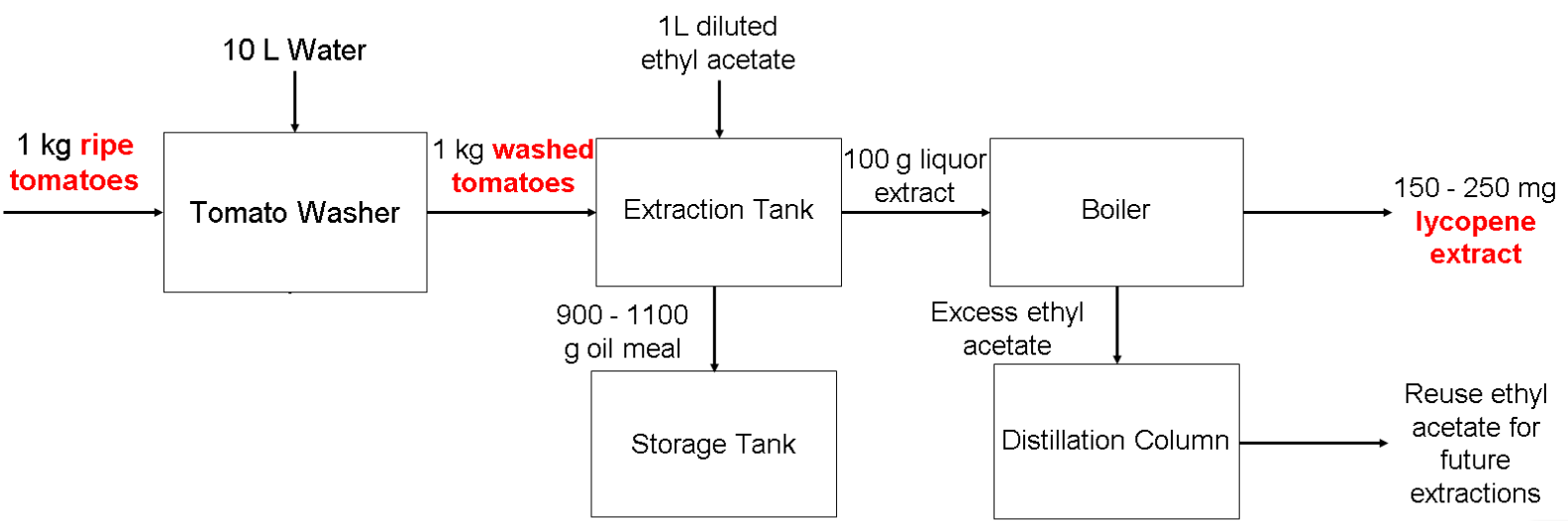
We then researched the operating parameters of each equipment in detail, citing our sources as well.
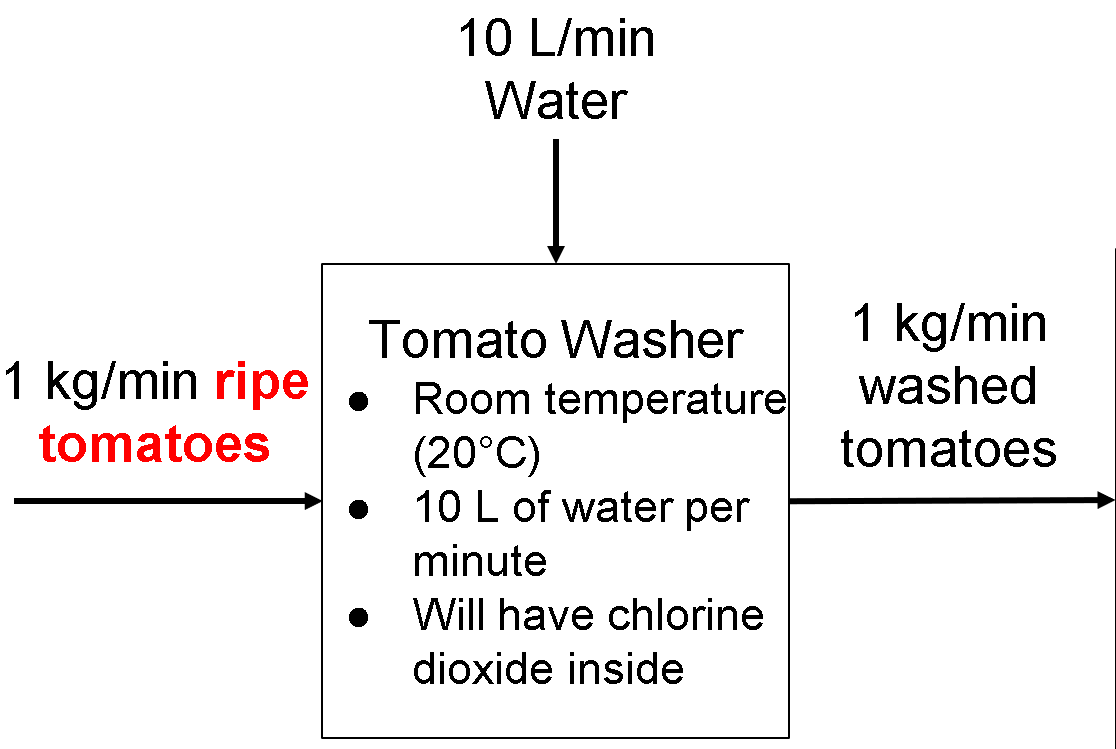
Tomato Washer Operating Parameters
Again, the tomato washer is used to clean the tomatoes to avoid contamination during the lycopene extraction process.
Temperature: The water will wash the tomatoes at a temperature similar to the tomatoes, which is room temperature. Using hot or cold water will create a temperature gradient between the water and the tomatoes, which will transfer not only heat but also microorganisms onto the tomatoes (https://www.bestfoodfacts.org/food-safety-produce/). Also, using room-temperature water avoids the need for a boiler or cooler for the water.
Flow Rate: We estimate roughly 10 liters of water is sufficient for every kilogram of tomatoes. 1 kilogram of tomatoes is about six medium-sized tomatoes weighing about six ounces each (http://www.tomatodirt.com/tomato-recipe-equivalents.html). We estimate 10L/min of water to flow through the tomatoes to wash off potential contaminants as well as loosen the tomato skins in making lycopene.
Composition: The water will not contain any soap or detergent because it could stick to the tomatoes and potentially contaminate the lycopene during the extraction process. (https://nifa.usda.gov/sites/default/files/resource/Guide%20to%20Washing%20Fresh%20Produce508.pdf). But, the water will contain 5 parts per million of chlorine dioxide (ClO2) to help decrease salmonella bacteria concentration on the tomatoes by 5 orders of magnitude (http://ezproxy2.library.drexel.edu/login?url=https://search-proquest-com.ezproxy2.library.drexel.edu/docview/231328537?accountid=10559)
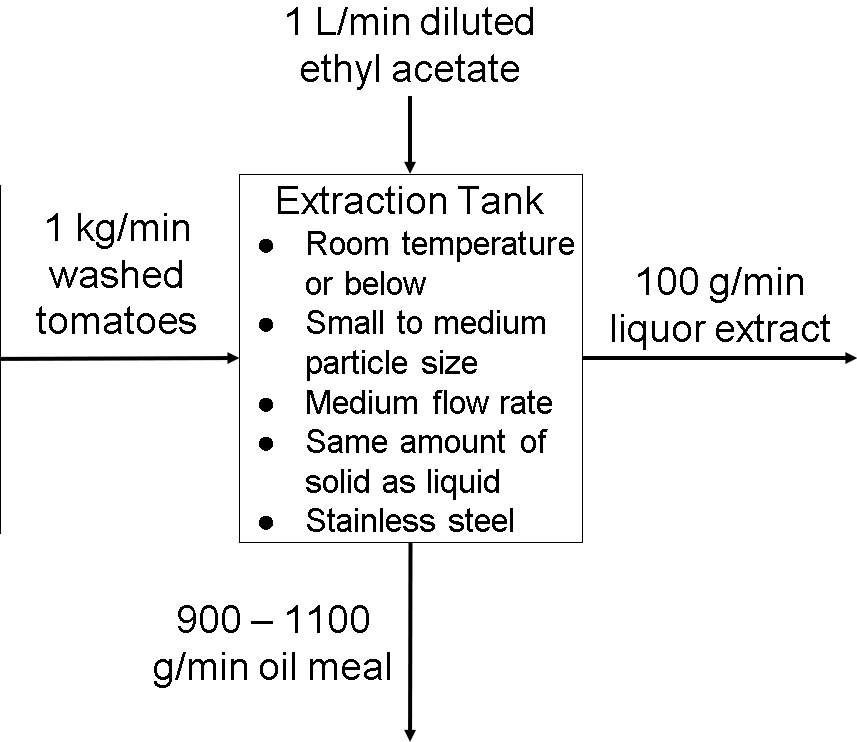
Extraction Tank Operating Parameters
The extraction tank is used to extract liquor extract from the washed tomatoes.
Temperature: The best temperature to extract lycopene is at room temperature or below. We don’t want the temperature to be too hot because it could degrade and denature the lycopene inside the tomatoes. Cooking temperatures above 100°C will ruin the lycopene and it will become unstable (https://ucanr.edu/datastoreFiles/608-1104.pdf). We don’t want to extract it when the tomatoes are frozen either because then it will be hard and slow to separate them. This is supported by our temperature experiment. The low temperatures did not extract a lot of coffee from the coffee grounds, resulting in a low coffee extract concentration. The highest temperature we tested extracted the most though, but this doesn’t mean that higher temperatures, like 100°C, will have a higher coffee concentration. The coffee might be extracted too fast that the extraction will just have water pouring out after that, making the coffee concentration very low in total.
Particle Size: Particle size will depend on how much lycopene we want to extract since one small, ripe tomato only has about 1-8 mg of lycopene. Since we are not actually doing a lycopene extraction, a small to medium particle size will be best for the extraction tank. The tank won’t be able to handle large or very small particle sizes because it might take too long to extract from large tomatoes, or not enough to extract from the very small tomatoes. There wouldn’t be too much or not enough ethyl acetate for the tomatoes too. This claim can be supported by our coffee experiments. We realized that it took too long to grind 10 grams of coffee beans per trial every week, so we changed it to 5 grams. We didn’t want to lower it too much because then there wouldn’t be enough coffee grounds to test out one trial (there were three trials for every experiment we did).
Flow Rates: The flow rates should essentially be the same as the two other equipment after the extraction tank since we don’t want the whole process to malfunction. However, it also depends on how long the extraction from the tomatoes will take. We want the extraction to be accurate too, making little to no mistakes. A medium flow rate would be best for the extraction tank since it is going to take a decent amount of time to extract from one tomato. If we chose a low or high flow rate, the extraction tank may be too slow or too fast for the other equipment to handle, and may not extract all of the liquor extracts from the tomatoes.
Solid:Liquid Ratio: Since the extraction tank will have 1 kg of ripe tomatoes and 1 L of ethyl acetate, the solid to liquid ratio should be 1 kg/L. Again, if we increased or decreased the amount of tomatoes or ethyl acetate, then the latter should also be changed to extract the most lycopene efficiently. This can be supported by our solid:liquid ratio experiment. Since we wanted to extract the most amount of coffee for each trial, we kept the mass of coffee beans as known masses, but not the volume of water. This is how we determined how much water was needed for each mass of coffee beans to extract efficient coffee concentrations for each trial.
Composition: Stainless Steel (https://www.gsctanks.com/about-us/). The benefits of stainless steel are that it keeps the temperature inside efficiently and it is very durable to the point where it can be in good shape for years and years without repairs. It is also water-resistant and does not rust easily. However, stainless steel is very expensive and heavy. That is why not everyone can afford it despite all the benefits it has. (https://sciencing.com/advantages-disadvantages-stainless-steel-appliances-6088439.html). Without any research, we also think stainless steel is a good choice for the extraction tank based on our coffee experiments, specifically the filtration method experiment. Since the coffee filter paper gave a better coffee extract concentration than paper towels, this tells us that the materials used should be efficient with the type of extraction processed. The coffee filter gave better results because it is designed to filter coffee. Paper towels are not really designed to filter coffee. Since stainless steel can last for a very long time and store temperature, we would say that it is a good composition choice. If we were to choose other types of composition for the extraction tank, they would not be as heat-concealing and long-lasting as stainless steel, but they would be cheaper.
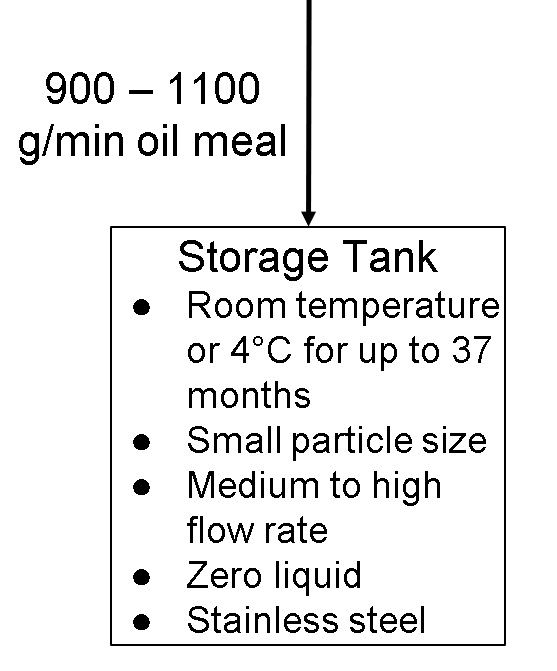
Storage Tank Operating Parameters
The storage tank is used to hold the by-products (oil meal) of the tomato extraction with some ethyl acetate since they are easily separable.
Temperature: Room temperature or 4°C for up to 37 months (http://www.fao.org/fileadmin/templates/agns/pdf/jecfa/cta/71/lycopene_extract_from_tomato.pdf). These temperatures make the oil meal stable when it’s stored in the storage tank. Any temperature that is higher or lower than the ones specified will spoil the oil meal sooner. We did not experiment with this, but we stored our coffee beans at room temperature at our house. We did not put it in the refrigerator or anywhere that is hot because coffee beans can already preserve well at room temperature as it is dry.
Particle Size: The particle size that the storage tank can hold doesn’t matter, but since oil meal that comes from the extraction tank will most likely be in powder form, we can say that small particle sizes will be in the storage tank. This is also supported from our particle size experiment, where we tested three different particle sizes (fine, medium, coarse) to see which one would extract the most amount of coffee. As expected, the fine particle size extracted the most amount of coffee. If we chose medium or coarse particle sizes for the storage tank, then the tank won’t hold as much as if it was holding small particle sizes.
Flow Rates: The flow rate really depends on the size of the storage tank. If it is a small storage tank, then the flow rate would have to be low so that the tank doesn’t overfill easily. If it is a medium-sized tank, then the flow rate would be medium, and if it is a large tank, then the flow rate would be high. It also depends on how fast people want the tank to fill up and how fast the overall process is. There’s nothing that we researched to support our claim, but we believe that a medium to high flow rate into the tank would be best (assuming the storage tank is large). Again, if we chose a low flow rate, then the oil meal might spill between the extraction tank and storage tank because they both have to be the same flow rate. If we chose a very fast flow rate, then the storage tank may fill up too fast and overflow.
Solid:Liquid Ratio: There isn’t really a solid:liquid ratio for the storage tank since all it’s doing is storing small particle-sized, dried oil meals and no liquids.
Composition: Stainless Steel (https://www.gsctanks.com/about-us/). Again, the benefits of stainless steel is that it conceals temperature inside efficiently and it is very durable to the point where it can be in good shape for years and years. It is also water resistant and does not rust easily. However, stainless steel is very expensive. That is why not everyone can afford it despite all the benefits it has. (https://sciencing.com/advantages-disadvantages-stainless-steel-appliances-6088439.html). We also think stainless steel is a good option for the storage tank because since the temperature has to be 4°C to room temperature for up to 37 months, the composition can retain that temperature easily and for a long time too, so the oil meal will be in good condition for a long time. During our coffee experiments, we saw that the plastic or glass cups we used did not retain the temperature we started off with, decreasing by 2°C to 3°C every cup. Therefore, we would need a material that would retain temperature efficiently, so the best option would be stainless steel for the storage tank. If we were to choose any other type of composition, they would not retain heat inside very well, and they might not be sturdy and durable. The only benefit of using other compositions is that they would be cheaper than stainless steel.
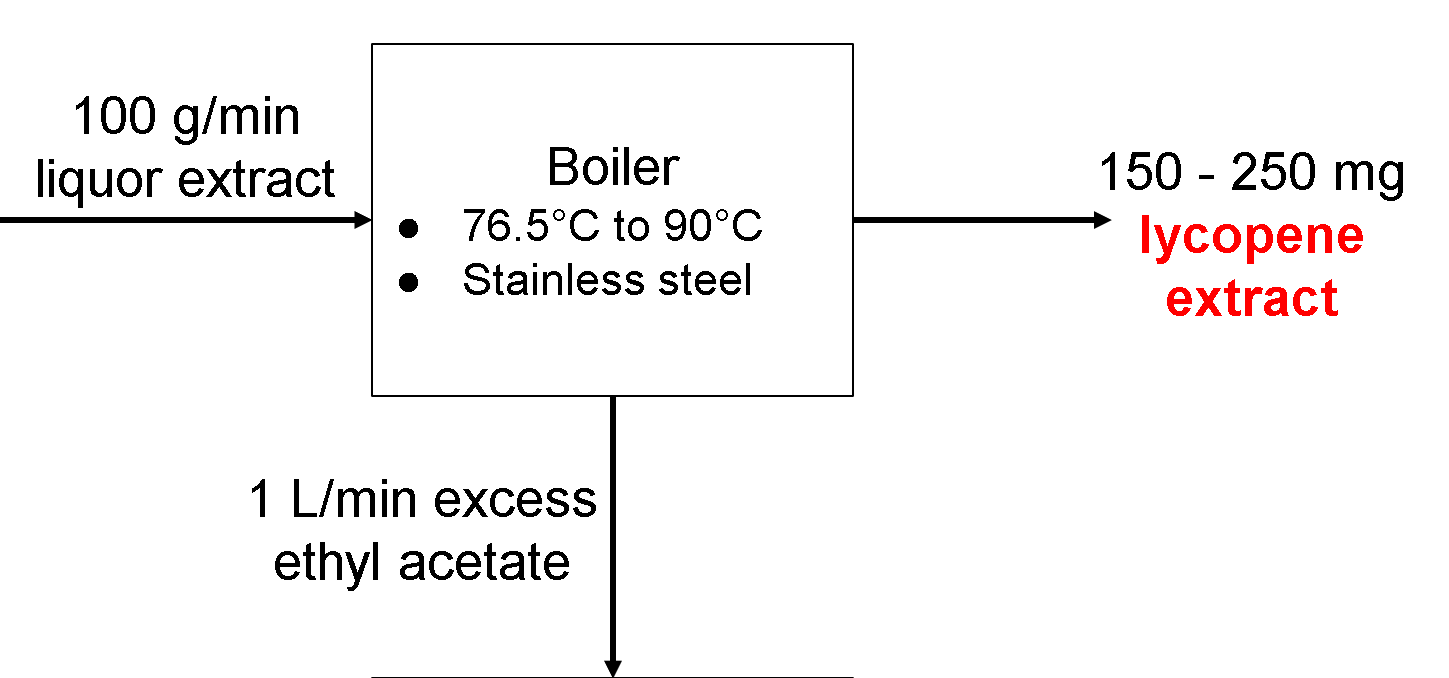
Boiler Operating Parameters
The boiler is used to boil the liquor extract to a certain temperature in order to separate ethyl acetate from the liquor extract to produce pure lycopene extract.
Temperature: At least 76.5ºC, the boiling point of ethyl acetate, and at most 90ºC, the denature temperature for lycopene. Higher temperatures will boil off the ethyl acetate faster but may pose a greater fire risk (higher temperatures would increase the vapor pressure of ethyl acetate and may make it easier to ignite. The flashpoint of ethyl acetate is -3.3ºC, so we have to be careful even before that (https://www.sigmaaldrich.com/chemistry/solvents/ethyl-acetate-center.html). Since the point of this extraction step is to essentially dry off the final amount of ethyl acetate from our lycopene, it is important that our temperature is high enough to boil off the ethyl acetate without accidentally altering the final amount of lycopene that is obtained in our extraction.
Composition: The boiler will be made of Stainless Steel due to its low specific heat capacity (http://asm.matweb.com/search/SpecificMaterial.asp?bassnum=mq304a). This, along with its high structural integrity allow it to be a viable material for our boiler to be made out of since it will be able to withstand the changes in temperature that we will be aiming for. Stainless steel is also highly resistant to rust, which may be a problem when using other metals such as iron or other steel alloys. If it does not rust, then no contaminants related to rusting will get into our final product and keep it pure as a result.
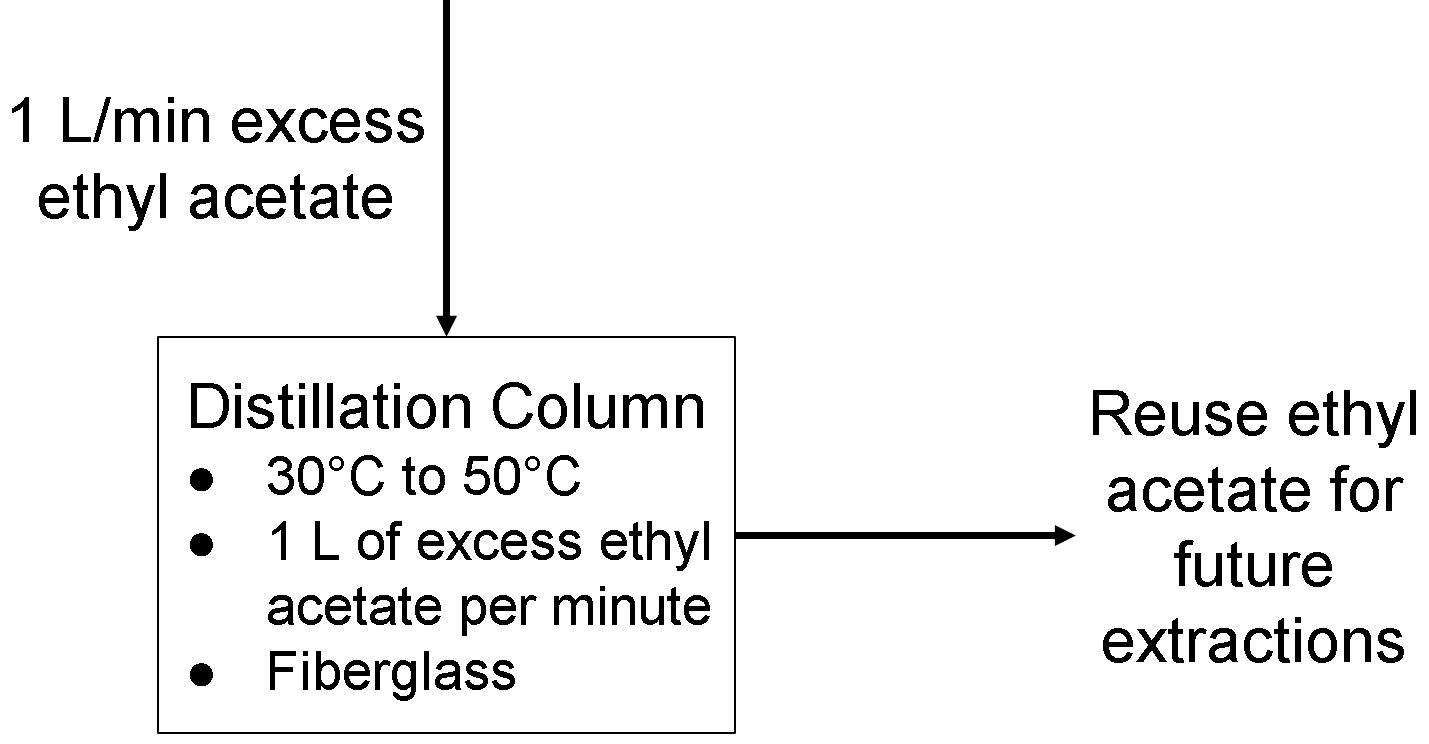
Distillation Column Operating Parameters
The distillation column is used to collect the excess ethyl acetate that does not react and is removed from the boiler equipment. This would help with reducing the amount of waste that this process will inevitably produce. We can also reuse the ethyl acetate for future extractions.
Temperature: As the boiling point of ethyl acetate is 76.5°C, the distillation column should be below that at least a few degrees, preferably 20°C less to make the distillation process quicker, so around 30-50°C should be okay.
Flow Rate: The flow rate of excess ethyl acetate into the distillation column cannot be too high. As flow rate increases for a binary separation, the purity of the collected substance (ethyl acetate here) decreases (https://controlguru.com/distillation-introduction-to-control/). If the flow rate is too low, then the distillation column might not keep up with other parts of the PFD (more ethyl acetate is coming in than the distillation column can process). We estimate the flow rate to be at most 1 liter/minute in accordance with the amount of materials on our PFD.
Composition: A fiberglass composite, such as glass with polyester resin to make a fiberglass-reinforced polymer (FRP), will make up the distillation column. This FRP is able to handle the temperature operating range while also being relatively rigid and corrosion-resistant compared to other metals or plastics. This FRP also has self-extinguishing flammability, making it relatively reliable and long-lasting (https://www.automation.com/en-us/articles/2015-2/comparing-stainless-steel-polycarbonate-and-fiberg).
Final Lycopene Extraction PFD with Optimal Operating Parameters
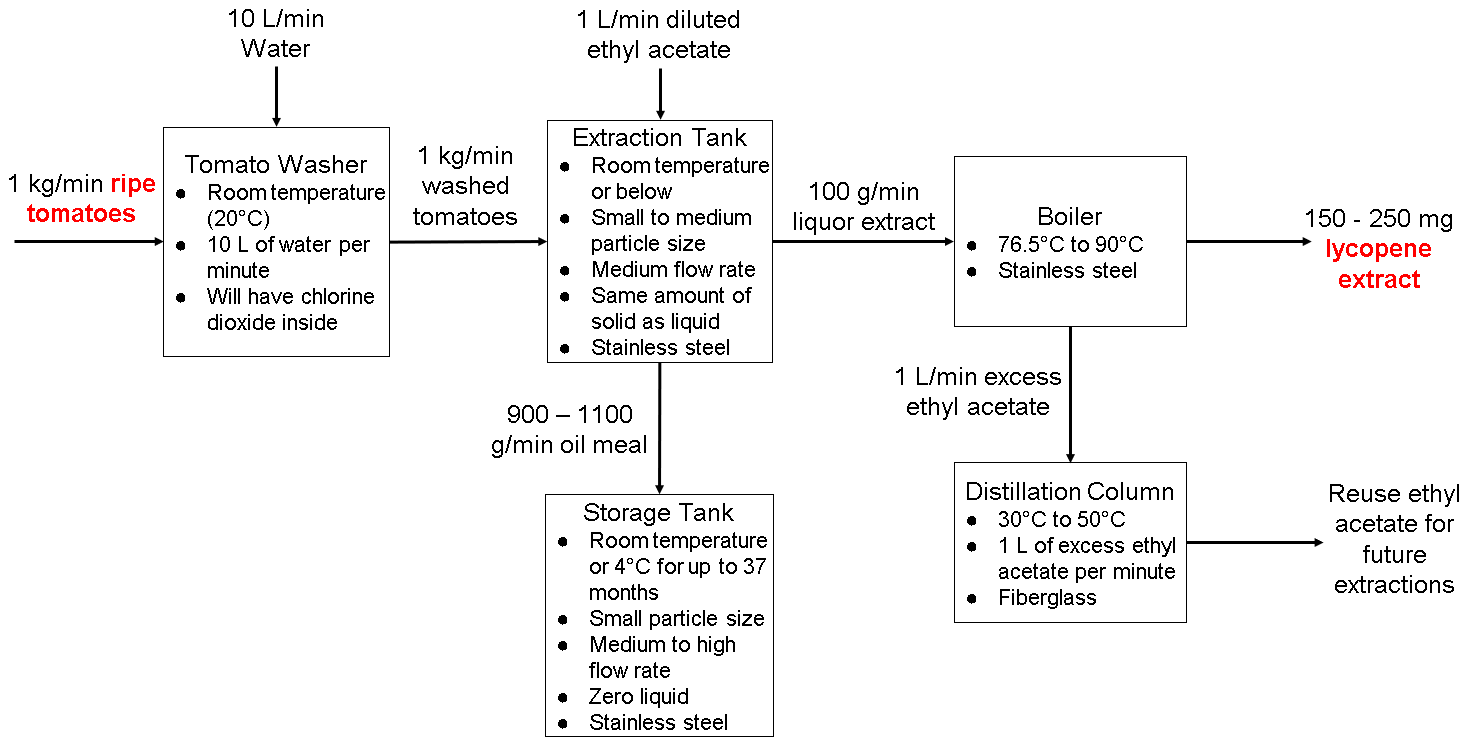
We will insert this information into our final report. The report will also talk about our weekly experiments with each operating parameter, and how our results relate to lycopene extraction.